- Registration Service of Medical Devices
- CRC services Medical Device Business License Medical Device Registration Contract Research Organization ISO 13485 Quality System Certification GMP CE Certification Service FDA Registration (Filing) Software Integrity Verification Service Production process validation service ISO 15378 Medical Package System Certification ISO 13485 Internal Auditors Training Regulatory Services For Medical Devices
- Information security service of medical
- ISO 27001 Information Security Certification ISO 20000 Information Service Certification Information system grade protection filing Business Continuity Management Services Internal Auditor Training
- Medical Software Development
- Software of Good Supply Practice(GSP) Production System Development Customized software development
- CONTACT US
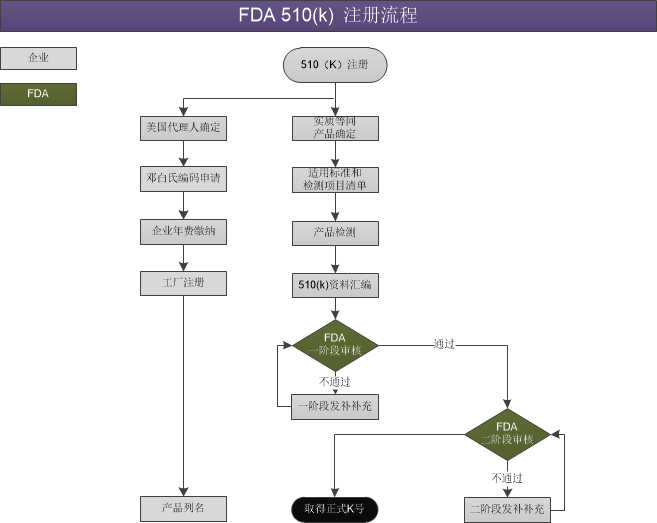
FDA 510(k) 注冊流程
CREATE TIME:2018-06-29 11:34BROWSE TIMES:2761
FDA是食品藥品監(jiān)督管理局(Food and Drug Administration)的簡稱。FDA有時也代表美國FDA,是國際醫(yī)療審核權威機構,由美國國會即聯(lián)邦政府授權,專門從事食品與藥品管理的最高執(zhí)法機關。
對于生產(chǎn)II類醫(yī)療器械的生產(chǎn)商來說,需要在美國食品藥品管理局(FDA) 申請市場準入,即FDA 510K申請。只有通過美國FDA注冊認證,才能獲得在美國合法的銷售許可。
PREVIOUS: NOTHING
NEXT: NOTHING
NEXT: NOTHING
